Research Theme
5.Lipidomics of glucosylated lipids in the CNS
Kazuki Nakajima and Yoshio Hirabayashi
Laboratory for Molecular Membrane Neuroscience, RIKEN Brain Science Institute
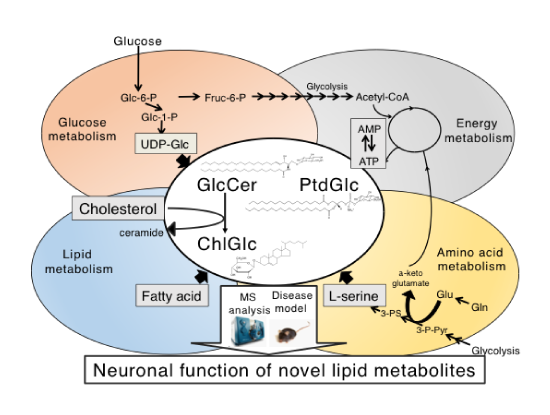
Glucose is the most important component for living organisms. Brain is the metabolically active tissue in animals and depends exclusively on glucoses as energy source to produce adenosine triphosphate (ATP). Glucose is also used for endogenous synthesis of membrane lipids such as fatty acid and cholesterol. Moreover, it is metabolically converted to L-serine, which serves as a precursor amino acid for the synthesis of sphingolipids including sphingomyeline and glycosphingolipids. Thus, it could be said that glucosylceramide (GlcCer), one of characteristic membrane glycolipid in the CNS is generated from three major metabolic systems, glucose, amino acid (L-serine), and lipids (fatty acid) with highly coordinated manner at the organismal level [1-3]. Thus, it is quite reasonable to think that glucosylceramide and its related lipids play a fundamental role in metabolism homeostasis. For example, intracellular levels of GlcCer are tightly regulated by a crucial cellular energy sensor, AMP-activated protein kinase (AMPK) [4]. Recent our work has also shown that GlcCer synthase in Drosophila fat body is involved in the regulation of energy (glucose and lipid) metabolism [5]: overexpression of the enzyme in the fat body increases levels of stored nutrients such as triacylglycerol, glycogen and carbohydrate, whereas reduced expression of the enzyme results in a reduction of triacylglycerol levels.
To date, in addition to the GlcCer, two glucosylated lipids, phosphatidylglucoside (PtdGlc) and cholesterylglucoside (ChlGlc) have been identified in mammalian cells and tissues. Intriguingly, these three glucosylated lipids are enriched in lipid rafts/lipid microdomains, indicating that lipid glucosylation plays fundamental roles in a variety of cellular processes. PtdGlc is a minor glycerol-glycolipid species found in developing brains, and have the mass number identical with phosphatidylinositol [6, 7]. Very recently we have demonstrated that water-soluble lyso-form of PtdGlc repels TrkA-positive axons of DRG neurons at very low concentrations via G-protein coupled 7-transmembrane receptor, GPR55 [8]. On the other hands, ChlGlc, a member of the glucosylated sterols, was synthesized from GlcCer and cholesterol in UDP-glucose independent manner [9]. Recently, we identified β-glucosidase 1 (GBA1) as the enzyme responsible for ChlGlc formation in mammalian cells [10]. Very interestingly, dysfunction of GBA1 is known to be a high risk factor of Parkinson’s disease [11, 12].
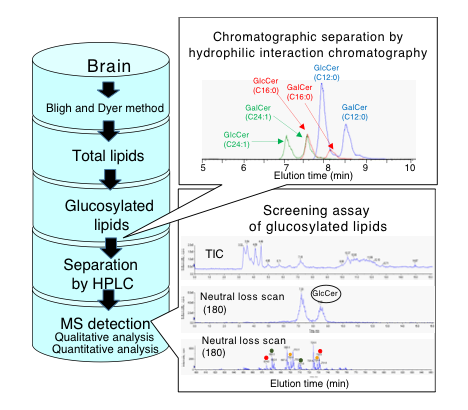
In order to grasp dynamic metabolic crosstalk in glucosylated lipid synthesis and their neuronal functions in response to pathological conditions, we proposed a functional lipidomics focusing on the glucose-related lipids. The lipidomics include the identification of biosynthetic and catabolic enzymes, and receptor, if there exists, for each glucosylated lipids, and novel lipid metabolites. Recent development of mass spectrometry enables us to identify and quantify even extremely minor novel lipids in brain tissues. One of the our challenging researches for monitoring glucosylated lipids is to develop chromatographic method to separate lipids that are not distinguished by regular MS alone, because abundant lipids having identical MS number exist in brains. We are trying to develop a novel target lipidomics approach for efficiently detecting such lipid species, which includes chromatographic separation techniques in lipid or gas phase, and screening assay system by triple quadrupole MS [13].
【References】
[1] Ishibashi Y et al. Biochim Biophys Acta., 1831(9):1475-85 (2013)
[2] Hirabayashi Y. Proc Jpn Acad Ser B Phys Biol Sci., 88(4):129-143 (2012)
[3] Hirabayashi Y et al. Seikagaku., 86(6):735-743 (2014)
[4] Ishibashi Y et al. J Biol Chem., 290(29):18245-18260 (2015)
[5] Kohyama-Koganeya A et al. J Lipid Res., 7, 1392-1399 (2011)
[6] Nagatsuka Y et al. FEBS Lett., 497(2-3), 141-147 (2001).
[7] Nagatsuka Y et al, Proc Natl Acad Sci U S A., 100(13):7454-7459 (2001)
[8] Guy A et al. Science., in press.
[9] Akiyama H et al. Biochim Biophys Acta., 1811(5):314-322 (2011).
[10] Akiyama H et al. Biochem Biophys Res Commun., 441(4):838-843 (2013).
[11] Sidransky E et al. N Engl J Med., 361(17):1651-1661 (2009)
[12] Mazzulli JR et al. Cell., 146(1):37-52 (2011)
[13] Nakajima K et al. Glycoscience: Biology and Medicine., 103-110 (2014).
